4DMedical receives CE Mark for CT:VQ™, enabling commercial launch across the European Union

4DMedical Limited (ASX:4DX), the global leader in cardiothoracic imaging software, announces that its latest imaging technology, CT:VQ™ has received CE Mark certification for commercial use in the European
Union. 4DMedical will quickly launch commercial deployment of CT:VQ across one of the world’s largest respiratory imaging markets.
Not Just for Smokers: The Surprising Faces of Lung Disease

We may all be guilty of picturing the same person when we hear that someone has lung cancer—an older man who smoked for many years. The truth is, millions of people with lung disease have never smoked a single cigarette. Children, women who have never smoked, and even athletes can suffer from life-altering respiratory disorders, too.
Lung Imaging in 2026: How Functional CT and New Investments Are Changing the Game
Lung disease is a global health challenge. Conditions like COPD, emphysema, and lung cancer affect hundreds of millions of people worldwide, yet many patients are still being diagnosed too late. Traditional imaging has focused on structure: identifying nodules, masses, or obvious tissue changes. In 2026, the field is rapidly evolving toward functional insights, understanding not only what the lungs look like but also how they function, region by region. Recent research, industry debate, and major investment activity all point to a pivotal moment in pulmonary imaging.
4DMedical Secures US$100+ Million Funding to Accelerate U.S. Expansion and increase Technology Dominance in Software-Based Lung Imaging

Funding will scale hospital deployments for groundbreaking CT:VQ™ imaging product, strongly advance R&D, support early adopters at Stanford, Cleveland Clinic, University of Miami, UC San Diego Health, and more
4DMedical, the leader in software-based respiratory imaging technology, today announced US$100+ million (AU$150 million) in new institutional placement. The investment will primarily be leveraged to accelerate U.S. adoption of its class-leading “CT:VQ™” product and significantly boost research and development—bolstering 4D’s technology lead in software-based lung imaging.
Aunt Minnie: 4DMedical inks major distribution agreement for CT:VQ™

New coverage from AuntMinnie.com: Philips will distribute 4DMedical’s CT:VQ™ across healthcare systems in the U.S. and Canada, expanding access to non-contrast CT-based ventilation and perfusion mapping.
Catching COPD Early: Why Timing Could Save Your Lungs
Chronic Obstructive Pulmonary Disease, or COPD, is one of the most common lung conditions in the world—yet it remains deeply underdiagnosed. In the United States alone, over 12.5 million people are known to have COPD, but millions more may be living with it. One of the main reasons for this gap is that early symptoms, such as a lingering cough or shortness of breath, are often easily dismissed. Many people don’t seek help until their breathing problems are severe and irreversible damage has already taken place.
CT:VQ™ Redefines Lung Imaging: FDA Clearance Makes Non-Contrast V/Q Scans a Reality
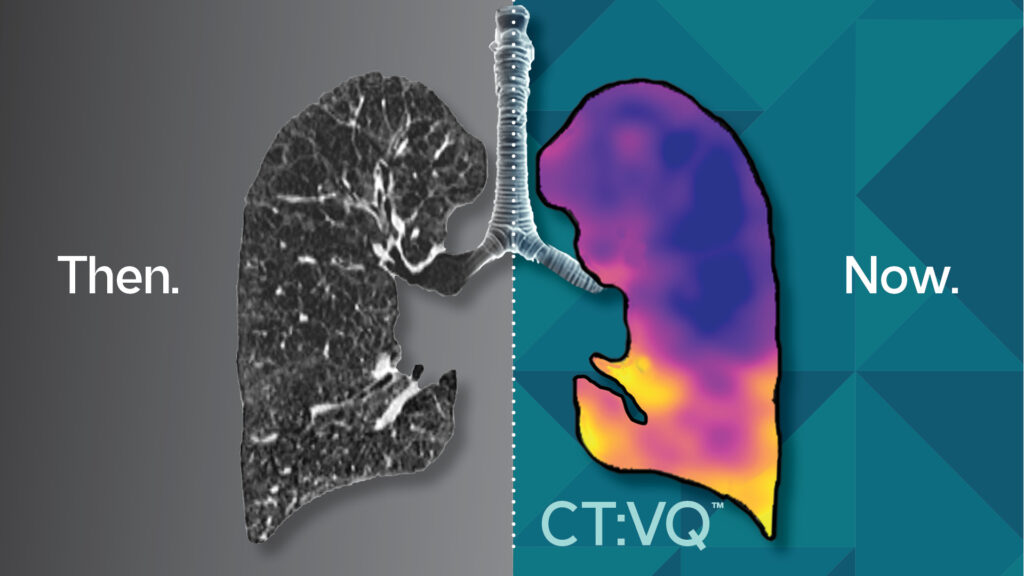
In a landmark move for pulmonary diagnostics, 4DMedical’s CT:VQ™ earned FDA 510(k) clearance in September 2025, marking it as the first and only non-contrast ventilation–perfusion (V/Q) imaging solution. The technology is available via routine chest CT scans. With CMS confirming reimbursement under Category III CPT codes (on top of the existing chest CT payment), CT:VQ™ is poised to redefine accessibility and efficiency in functional lung imaging.
4DMedical’s CT:VQ™ Receives FDA 510(k) Clearance; First-and-Only Non-Contrast VQ Imaging
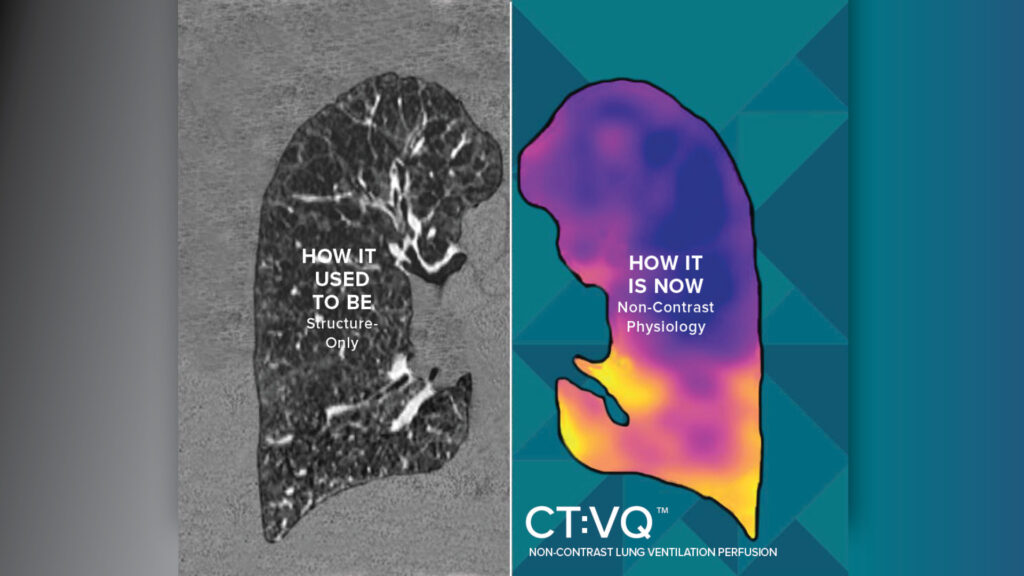
4DMedical, a leader in advanced respiratory imaging, today announces U.S. Food and Drug Administration (FDA) 510(k) clearance for CT:VQ™, the world’s first and only non-contrast, ventilation–perfusion (VQ) imaging solution. In parallel, the U.S. Centers for Medicare & Medicaid Services (CMS) has confirmed reimbursement for CT:VQ under Category III CPT codes; this payment is in addition to existing reimbursement for the underlying chest CT.
New Study Underscores XV Technology’s Power to Detect Hidden Lung Disease

A major new multi-center study published in Respiratory Research (July 2025) demonstrates that 4DMedical’s X-ray Velocimetry Lung Ventilation Analysis Software (XV LVAS®) can reveal early and subtle forms of small airways disease that are often missed by standard tests like spirometry and CT scans. Researchers from Vanderbilt University, Johns Hopkins, University of Miami, and Alfred Hospital in Melbourne showed that XV technology identifies disease-specific and severity-specific biomarker patterns in chronic obstructive pulmonary disease (COPD) and deployment-related constrictive bronchiolitis (DR-CB)—even when conventional tests appear normal.
Innovative XV Technology Shows Promise for Detailed Lung Function Assessment in Bronchiectasis Patients
A new study led by UNSW Sydney researchers demonstrates the feasibility and promise of the innovative XV Scanner™ with XV LVAS®, non-invasive imaging tools developed by 4DMedical, for assessing regional lung function in adults with bronchiectasis caused by Primary Ciliary Dyskinesia (PCD) and other conditions.
